BOARDS OF APPEAL
Decisions of the Technical Boards of Appeal
Decision of Technical Board of Appeal 3.3.1 dated 27 June 2003 - T 1020/98 - 3.3.1
(Translation)
Composition of the board:
Chairman: | A. J. Nuss |
Members: | R. Freimuth |
| R. T. Menapace |
Applicant: Bayer CropScience GmbH
Headword: Safeners/BAYER
Article: 84, 111(1) EPC
Keyword: "Clarity (yes) - complex claim not unclear - within reasonable time - Markush formula - hierarchical structure of substituent definitions" - "Conciseness (yes) - Markush formula the most concise formulation of a class of chemical compounds - greater ease of substantive examination not determinative of the content of an independent claim"
Headnote:
I. Compliance with the clarity requirement of Article 84 EPC is not dependent on the time required to establish whether a given compound is covered by the product claim. The clarity requirement is not a basis for objecting to the complexity of a claim, as complexity is not tantamount to lack of clarity. The simplicity of an individual claim is not a criterion for the granting of a patent under the EPC.
II. A Markush formula is the concisest means of defining a class of chemical compounds in a claim.
III. There is no legal basis in the EPC for a request to restrict the content of an independent claim so that substantive examination can be carried out with greater ease and less effort. Furthermore, under the EPC, making the examining division's work easier is not a prerequisite for starting substantive examination of a patent application.
Summary of facts and submissions
I. The appeal, received on 15 July 1998, lies from the examining division's decision posted on 5 May 1998 refusing application No. 93112074.5 (publication No. 582 198).
II. The examining division found the application to be in breach of the requirements of Article 84 EPC. The basis for its decision was a main request comprising claims 1 to 14 as amended in a submission dated 20 March 1997, independent claims 1 and 5 of which read as follows:
"1. Use of compounds of formula I or salts thereof
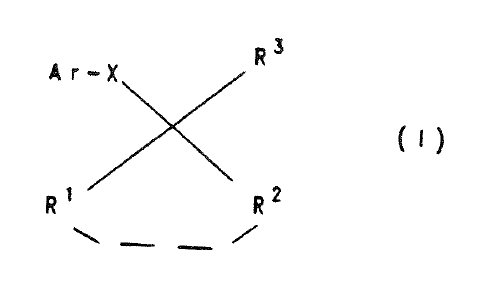
wherein:
R1 and R2 independently are radicals of the formula
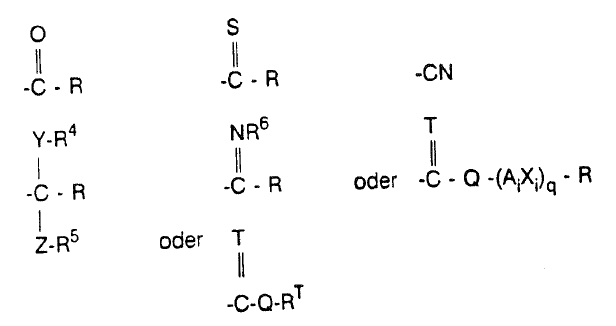
wherein R, RT, R4, R5, R6, Y, T, Z, Q, Ai, Xi and q are as defined below; or R1 and R2 are joined and together form a group of the formula -CO-Q1-D-Q2-CO-
wherein:
Q1, Q2 independently are defined as for Q and
D is a bivalent group of the formula CR'R'' or C=O, where R' and R'' are independently hydrogen or C1-C4-alkyl;
R3 is hydrogen, halogen, C1-C18-alkyl, C2-C8-alkenyl, C2-C8-alkynyl, C1-C18-alkoxy, C2-C8-alkenyloxy, C2-C8-alkynyloxy, C1-C18-alkylthio, C2-C8-alkenylthio, C2-C8-alkynylthio, each of the last nine radicals individually being unsubstituted or substituted by one or more radicals selected from halogen, nitro and cyano; or C3-C12-cycloalkyl unsubstituted or substituted by one or more radicals selected from C1-C4-alkyl, halogen, nitro and cyano; or SiRaRbRc wherein Ra, Rb and Rc are independently C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl or unsubstituted or substituted phenyl; or a radical of the formula Ar'X'- wherein Ar' and X' are defined analogously to Ar and X respectively;
X is O, S, NH-NH or NRd, wherein Rd is defined analogously to R4, or -CH2O-, -CH2S-, -CH(Ar)O- or -CH(Ar)S-;
Ar is an aromatic radical;
R is hydrogen or an aliphatic, aromatic, heteroaromatic, araliphatic or heteroaraliphatic radical with from 1 to 30 C-atoms and one or more functional groups as appropriate;
RT is a radical of the formula -CO-R, -CS-R, -NRfRg, N=CRhRi or SiRaRbRc,
wherein R has the above meaning and Rf, Rg, Rh and Ri independently are hydrogen, C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, benzyl, phenyl or substituted phenyl, or Rf and Rg together with the N atom are a 5 or 6-member heterocycle which may further comprise up to two other heteroatoms selected from N, O and S and may be substituted by C1-C4-alkyl, and Ra, Rb and Rc independently are C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, phenyl or substituted phenyl;
Y and Z independently are oxygen, sulphur in its different oxidation states or -NRe, wherein Re is defined analogously to R4;
R4 and R5 are the same or different and independently are hydrogen, C1-C6-alkyl, C2-C6-alkenyl, C2-C6-alkynyl, (C1-C4-alkyl)-carbonyl, wherein each of the last four radicals is unsubstituted or is substituted by one or more substituents selected from halogen, C1-C8-haloalkoxy, nitro, cyano, hydroxy, C1-C8-alkoxy and C1-C8-alkoxy, wherein one or more, preferably up to three not directly bound CH2 groups are replaced by oxygen, and C1-C8-alkylthio, C1-C6-alkylsulphonyl, C2-C8-alkenylthio, C2-C8-alkynylthio, C2-C8-alkenyloxy, C2-C8-alkynyloxy, C3-C7-cycloalkyl, C3-C7-cycloalkoxy and amino, mono and di-(C1-C4-alkyl)-amino; or formyl; or SiRaRbRc, wherein Ra, Rb and Rc independently are C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl or unsubstituted or substituted phenyl; or C3-C8-cycloalkyl, C3-C8-cycloalkenyl, heterocyclyl with from 3 to 7 ring atoms, aryl, heteroaryl or arylcarbonyl, with each of the last six radicals being unsubstituted or substituted by one or more radicals selected from C1-C8-alkyl, halogen, C1-C8-haloalkoxy, nitro, cyano, hydroxy, C1-C8-alkoxy and C1-C8-alkoxy, wherein one or more, preferably up to three not directly bound CH2 groups are replaced by oxygen, and C1-C8-alkylthio, C1-C6-alkylsulphonyl, C2-C8-alkenylthio, C2-C8-alkynylthio, C2-C8-alkenyloxy, C2-C8-alkynyloxy, C3-C7-cycloalkyl, C3-C7-cycloalkoxy and amino, mono and di-(C1-C4-alkyl)-amino; or
R4 and R5 together are a C2-C4-alkylene chain or a C2-C4-alkenylene chain which is unsubstituted or substituted by one or two radicals selected from methyl, ethyl, methoxy, ethoxy and halogen;
R6 is hydrogen, C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, C6-C12-aryl, heteroaryl, benzyl, C1-C4-alkoxy, acyloxy, hydroxy, -NH-CO-NH2, -NH-CS-NH2, mono and di-(C1-C4-alkyl)-amino, -NH-acyl, -NHSO2-(C1-C4-alkyl), C6-C12-aryloxy, heteroaryloxy, NH-SO2-aryl or NH-aryl, wherein aryl or heteroaryl in the last four radicals is unsubstituted or is substituted by one or more radicals selected from halogen, nitro, (C1-C4)-alkyl, (C1-C4)-alkoxy, (C1-C4)-haloalkyl and (C1-C4)-haloalkoxy;
T is O or S, NR7, NOR7 or NO-acyl;
Q is O or S;
q is an integer from 0 to 4;
i is an index number which when q is not 0 takes on all integer values from 1 to q, q having the above meaning;
Xi independently equates to O, S, NR7, N-(Ai-Xi-)q-R;
Ai independently equates to unsubstituted or substituted C1-C6-alkylene, C2-C8-alkenylene, C2-C8-alkynylene, C3-C6-cycloalkylene, C3-C6-cycloalkenylene, heterocyclyl, aryl or heteroaryl; and
R7 independently equates to H, C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, C3-C6-cycloalkyl, C3-C6-cycloalkenyl, heterocyclyl, aryl or heteroaryl;
as safeners for protecting crop plants against phytotoxic side-effects of herbicides."
"5. Compounds of formula I or salts thereof as defined in one of claims 1 to 4, with the exception of:
(a) compounds of formula I, wherein:
Ar is phenyl substituted by the radicals U1, U2 and U3, where U1 is a radical selected from halogen, C1-C4-alkyl, C1-C4-alkoxy, CF3 and C1-C4-alkylsulphonyl and U2 and U3 are the same or different and are each selected from hydrogen, halogen, C1-C4-alkyl, C1-C4-alkoxy, CF3 and C1-C4-alkylsulphonyl;
X is an oxygen atom;
R1 is a group of the formula -COOR;
R2 is a group of the formula -COOR;
R3 is C1-C4-alkyl; and
R is identical or different radicals selected from hydrogen and C1-C4-alkyl
(b) compounds of formula I, wherein:
Ar is phenyl, 1,3-dichlorophenyl, 1,3,5-trichlorophenyl, 3-methoxyphenyl, naphthyl, coumarinyl, 4-methylcoumarinyl or 7-flavonyl;
X is an oxygen atom;
R1 is a group of the formula -COOR;
R2 is a group of the formula -COOR;
R3 is hydrogen; and
R is identical or different radicals selected from hydrogen, aryl, alkyl and aralkyl
(c) 2-(quinolin-8-yl-mercapto)-malonic acid diethyl ester,
2-(quinolin-8-yl-mercapto)-acetoacetic acid ethyl ester or
4-chloro-phenoxy-malonic acid diethyl ester."
III. The examining division gave the following grounds for the contested decision:
"(2) We take the view that for a claim defining compounds in a Markush formula to be deemed clear within the meaning of Article 84 EPC, it must be possible to establish with absolute certainty within reasonable time whether a given compound falls within the scope of the claim. Even for the use claims the situation is critical (although no disclaimers were necessary here as with regard to use no novelty-destroying prior art was to be found in the search report). Special problems are caused by the exceptional length of the claims, by the fact that the formula consists entirely of variables, and by the number of variables, mostly defined in terms of other variables.
(3) With regard to the product claims, the Article 84 situation is unacceptable. Given the considerable scope of a product claim (where, unlike a use claim, only the structure of the claimed compounds determines the scope, regardless of how they are used), the Article 84 requirements are particularly significant. The product claims here are formulated even more confusingly than the use claims (with two complicated Markush-style disclaimers to boot), and must be deemed in breach of Article 84.
(4) The disclaimers are held to be the main problem, as they attempt to establish novelty in an unnecessarily complicated and elaborate manner. They are therefore in breach both of the Article 84 requirement for claims to be formulated concisely and of the requirement for them to be formulated clearly. To give a clear idea of the scope of the product claims, novelty could easily have been established by deleting definitions of variables, which would also make it easier to establish the exact degree of overlap with a similarly complicated Markush formula.
(5) Since T 12/90, establishing the exact degree of overlap between two Markush formulae has become an important factor in assessing patentability. In the examining division's view, formulating claims in a style that makes routine tasks in substantive examination unnecessarily difficult is a contravention of Article 84 EPC."
IV. The appellant (applicant) with its statement of grounds of appeal on 15 September 1998 filed two further versions of claim 5 as auxiliary requests 1 and 2. It refuted the contested decision's assertion that claim 5 according to the main request was in breach of Article 84 EPC. In conjunction with claim 1 its wording did not pose any unusual obstacles likely to require undue effort during examination. For someone checking whether a given chemical compound was covered by claim 5, disclaimers (a) to (c) were clear and comprehensible. The formulation of the disclaimers was not complicated, but was derived from the cited publications. Nor, in the present case, could the disclaimers be replaced to comparable effect by a positive formulation.
The appellant requested that the contested decision be set aside and that a patent be granted on the basis of claims 1 to 14 according to the main request or of claims 1 to 14 with an amended claim 5 according to auxiliary request 1 or 2. Auxiliarily it requested oral proceedings.
Reasons for the decision
1. The appeal is admissible.
2. Extent of scrutiny in appeal proceedings
Article 111(1) EPC does indeed empower the boards of appeal to go beyond the grounds of the contested decision, exercising any power within the competence of the examining division which took that decision; but that does not mean they should conduct a full patentability examination of the application under appeal. That is the examining division's task. Even in ex parte proceedings, the procedure before the boards is primarily concerned with examining the contested decision (see G 10/93, OJ EPO 1995, 172, Reasons 4).
In the present case, the application in suit was refused solely because of the lack of clarity and conciseness of product claim 5. The board will therefore confine itself to reviewing and deciding whether claim 5 complies with these Article 84 requirements. It will not rule on whether the disclaimer in claim 5 is allowable in terms of the requirements of Article 123(2) EPC, that point of law being in any case the subject of Enlarged Board referrals G 1/03 and G 2/03 (OJ EPO 2003, 113).
Main request
3. Clarity (Article 84 EPC)
3.1 The contested decision objects to product claim 5 on the grounds that it lacks clarity because it defines the claimed compounds using a Markush formula and additionally using two "complicated" Markush-style disclaimers. The findings of fact that brought the examining division to this conclusion are not apparent from the statement of grounds for the decision.
The definition of the claimed compounds by means of a Markush formula results from product claim 5 referring back to use claim 1. The contested decision objects to the latter as being confusing and "critical", but does not dispute its clarity within the meaning of Article 84 EPC.
3.2 The board's first observation is that the structural description of chemical compounds by means of general Markush-style formulae is a classical method, part of the standard toolkit of the relevant skilled person, in this case a chemist. That has never been disputed.
In the present case the structure of the claimed chemical compounds is simply described in Markush formula (I), which comprises the five variable substituents Ar, X, R1, R2 and R3. The claim then defines these substituents by listing chemical groups for each of them, described by name or formula. Where the chemical groups are described in general formulae, they in turn comprise variable radicals, for each of which the claim gives a list of chemical groups. Thus, for example, for the variable substituent R3 of Markush formula (I) the claim gives a list of chemical groups including one with the general formula SiRaRbRc. According to the claim, its variable radicals Ra, Rb and Rc are in turn defined by a list of named chemical groups, namely C1-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl or unsubstituted or substituted phenyl. This structured, hierarchical format of the definition of the variable substituents Ar, X, R1, R2 and R3 of Markush formula (I) guides the rational skilled person through the claim and tells him clearly and unambiguously whether a given individual compound or class of compounds falls within the scope of the claim.
3.3 Further to the above Markush formula (I), product claim 5 includes the two disputed disclaimers (a) and (b) and a further undisputed disclaimer (c), the latter clearly referring to three individual chemical compounds (see point II above). The first two disclaimers exclude two precisely described compound classes from the subject-matter of the claim. These two excluded classes are defined by reference to Markush formula (I), with chemical groups identified by name or formula being specified for each of their variable substituents Ar, X, R1, R2 and R3.
In disclaimer (a), each of the substituents Ar, X, R1, R2 and R3 denotes only a single chemical group, Ar being a specifically substituted phenyl, X an oxygen atom, R1 and R2 a group of the formula -COOR, with radical R more closely defined, and R3 a C1-C4-alkyl. In disclaimer (b), the substituents X, R1, R2 and R3 likewise each denote only a single chemical group, X being an oxygen atom, R1 and R2 a group of the formula -COOR, with radical R more closely defined, and R3 hydrogen. The substituent Ar is defined in terms of a list of eight chemical radicals identified by name.
The challenged disclaimers (a) and (b) thus clearly and unambiguously define the two classes of compounds that are to be excluded from the subject-matter of the claim.
3.4 For these reasons the board concludes that the subject-matter of disputed product claim 5 is clearly defined by Markush formula (I) and disclaimers (a) to (c) and hence is clear within the meaning of Article 84 EPC.
3.5 The examining division based its contested decision among other things on the notion that for a claim to be deemed clear "it must be possible to establish with absolute certainty within reasonable time whether a given compound falls within the scope of the claim" (emphasis added by the board).
3.5.1 It is not apparent from the contested decision what is meant by the "reasonable time" yardstick, as there is no indication of a timescale or of any reasonability limit. Hence the grounds the examining division puts forward are unsound for lack of substantiation alone.
In the board's view, the actual time required does not really matter as long as the claim itself is clear. In the light of the observations under 3.2 and 3.3 above, that is the case here, as product claim 5 is hierarchically structured such that the skilled reader will require little effort to determine its subject-matter. Compliance with the clarity requirement as such is not dependent on the time required to establish whether a given compound falls within the scope of the product claim. That would make this purely quantitative time criterion an independent prerequisite for patentability for which there is no support either in the clarity requirement of Article 84 EPC - qualitative by its very nature - or anywhere else in the Convention.
3.5.2 This is in keeping with the decision in T 574/96 (not published in OJ EPO), which in Reasons 3.1 finds that the Article 84 clarity requirement offers no basis for objecting that a claim is not simple but complex and hence takes too long to understand, as complexity is not tantamount to lack of clarity of a claim. Under the EPC, the simplicity of an individual claim is not a self-contained requirement for a patent to be granted. Any such requirement would be inappropriate, as it would exclude from patent protection any invention having a subject-matter not describable in a simple claim formulation. Clarity within the meaning of Article 84 EPC merely requires the claims to define the subject-matter for which protection is sought clearly and unambiguously for the skilled person, if necessary in the light of the description. As set out above, that requirement is met in the present case.
4. Conciseness (Article 84 EPC)
4.1 The contested decision further objects to product claim 5 on the grounds that it lacks conciseness because its subject-matter is additionally defined using disclaimers referring to the claimed Markush formula (I), although novelty could also have been established by deleting meanings from the claimed lists of variable substituents.
4.2 The first thing to point out is that defining a class of chemical compounds by means of a Markush formula as in disputed product claim 5 is recognised as being the most concise way of formulating such subject-matter. Any other formulation, such as an individual listing of all encompassed subclasses or even of all encompassed individual compounds, would be lengthier and hence not so concise. This necessarily also applies to disclaimers (a) and (b), which refer to these Markush formulae and are structured accordingly.
The contested decision asserts that these disclaimers could easily have been replaced by deleting meanings from the claimed lists of variable substituents in the Markush formula, thereby producing an even conciser formulation of the subject-matter of the claim. This the board fails to follow, not least because no corroborating facts are given, and also because its own examination of the facts does not bring it to any such conclusion. Restricting the subject-matter of product claim 5 by deleting meanings from the claimed lists of variable substituents as requested by the contested decision does not yield the same scope as the present wording with disclaimers (a) and (b); it reduces the scope of the claim. For example, disclaimers (a) and (b) exclude oxygen as a meaning of substituent X. If that meaning were instead to be deleted from the claimed list of meanings for substituent X, all compounds with this meaning for X that are encompassed by Markush formula (I) would be excluded; whereas the existing formulation excludes only those compounds in which at the same time substituents R1 and R2 mean the group -COOR and substituent R3 means hydrogen or C1-C4-alkyl. The appellant's notice of appeal rightly makes explicit reference to these detrimental and unacceptable consequences for the scope of the claim.
In that light, product claim 5 in its present form is concise within the meaning of Article 84 EPC.
4.3 In its contested decision the examining division further based its request for an ultimately substantive restriction of the product claim on the notion that "formulating claims in a style that makes routine tasks in substantive examination unnecessarily difficult is a contravention of Article 84 EPC".
Article 84 EPC stipulates that the claims must define the matter for which protection is sought, must be clear and concise and must be supported by the description. To these exhaustive requirements for the claims the contested decision adds another, ie that the formulation of the claims must not make routine tasks in substantive examination unnecessarily difficult. The examining division is thereby seeking in effect to restrict the content of the product claim. Thus its request for a restriction of the content of an independent claim is linked to greater ease and less effort in substantive examination. Yet there is no legal basis in the European Patent Convention for this additional requirement that the examining division has sought to impose on the formulation of the subject-matter of an independent claim, even if its aim was to improve the efficiency of substantive examination. The requirement is therefore unlawful.
Consequently, an objection to a claim formulation under Article 84 EPC cannot be divorced from the requirement for clarity or conciseness, and based for example on the grounds that the formulation "makes routine tasks in substantive examination unnecessarily difficult". The applicant's duty is simply to comply with all the provisions of the EPC, including those designed to keep the effort required of the examining division in the grant procedure within reasonable bounds, whereas the examining division's duty, within the legal framework of the EPC, is to provide for optimum conduct of the proceedings, inter alia in terms of procedural economy, and also to ensure that the applicant complies with all provisions serving that purpose. Yet under the EPC a restriction of the content of an independent claim with a view to making the examining division's work easier is not a prerequisite for starting substantive examination of a patent application.
5. Remittal
The application in suit was refused solely for lack of clarity and conciseness of claim 5, and as the contested decision is not justifiable on those grounds, it must be set aside. The examining division has not yet taken an appealable decision on other issues concerning the patentability of all the claims, where concluding first-instance examination is still pending. The board therefore deems it inappropriate to decide on these issues in place of the first instance, in order to retain the possibility of examination by two instances in respect of these issues as well. In these circumstances the board, exercising its powers under Article 111(1) EPC, remits the case to the first instance for further prosecution.
Auxiliary requests
6. As the appellant's main request succeeds, there is no need for further examination of the subordinate auxiliary requests 1 and 2 or for the oral proceedings requested auxiliarily.
Order
For these reasons it is decided that:
1. The decision under appeal is set aside.
2. The case is remitted to the first instance for further prosecution on the basis of claims 1 to 14 according to the main request.